
W.H. Dudok van Heel and J.D. van der Toorn (1988)
A biological approach to water purification: II.
A practical application: The Delfinaario in Tampere, Finland
From: Aquatic Mammals 14(3): 92-106
Ozone can be generated by electrical discharge or by ultraviolet light. Photozone equipment(1) makes use of ultraviolet light to create a mixture of activated oxygen and ozone (McGregor, 1986). This process has been applied to ozonation of drinking water, in fish hatcheries and in wastewater treatment plants. One of the advantages of using ultraviolet light for the generation of ozone is that the amount of toxic by-products, such as nitrogen oxides is negligible In the Delfinaario, Photozone was designed to be used in two places in the system: with the foam fractionators, because it lowers the surface tension and thus facilitates foam formation (Schlesner, 1973) and as a final treatment of the water returning to the pools from the mixing channel. However, because of the high initial building costs, it was decided not to install the large Photozone unit that would treat the water going back to the pools. The units for the foam fractionators were installed as planned. These were units of the type PH 190 HD, which had one UV-lamp each.
The water in the Delfinaario is an artificial sea water mix, based on the GP2 medium, described by Spotte et al (1984). This mix has a salinity of 3.5%, a pH of 8.2 and an alkalinity of 2.8 meq/l. However the mix didn't turn out completely the way it was planned with respect to the trace metals. As can be seen from table I, many of the trace metals are present in concentrations that are much higher than was intended. This was the result of impurities in the bulk salts, like NaCl, MgCl2, CaCl2 and Na2SO4. Furthermore the fresh water obtained from the town water supply contained high levels of those elements, especially copper, zinc and manganese.
Possibly because of this imbalance it has been very hard to maintain the intended pH of 8.2. In November 1985 we tried if we could keep the pH at 8.2 by adding sodium hydroxide on a daily basis. At first it worked, but it became increasingly difficult. More and more hydroxide was needed. At first about 2.5 kg was added daily, later on that increased to as much as 6 kg per day. This strategy also had a very disturbing side effect : it increased the turbidity dramatically. After a month the visibility in the water was reduced to less than 3 meters. Laboratory tests done on the water indicated that this was due to the formation of colloidal solutions of calcium and magnesium hydroxide. It was then decided to discontinue the addition of hydroxide and let the system balance itself.
This new strategy resulted in the dissolution of the hydroxide particles and a consequent increase in water clarity. It then turned out that the ideal situation for our system was a pH between 7.8 and 8.0. Now that the system is balanced the pH slowly drops from about 8.0 to about 7.8. At the same time the alkalinity drops from about 3.0 to about 2.2. The drop in alkalinity is linear : 0.061 meq/l/day (r=-0.997). To bring the alkalinity back up from 2.2 to 3.0, about 100 kg of anhydrous sodium bicarbonate must be added. This addition is necessary every 15 to 16 days.
| Species | Units | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| Cl- | mg/l | 18800 | 19300 | 23600 | 18000 |
| SO42- | mg/l | 2715 | 2712 | 2800 | 1800 |
| Na+ | mg/l | 10700 | 10800 | 11500 | 11600 |
| Mg2+ | mg/l | 1290 | 1290 | 1020 | 730 |
| Ca2+ | mg/l | 412 | 409 | 360 | 400 |
| K+ | mg/l | 380 | 399 | 391 | 430 |
| Fe3+ | µg/l | 2 | 195 | 60 | 25 |
| Zn2+ | µg/l | 4.9 | 4.97 | 560 | 260 |
| V | µg/l | 2.5 | 2.55 | 60 | 350 |
| Mn2+ | µg/l | 0.2 | 0.198 | 60 | 60 |
| Cu2+ | µg/l | 0.5 | N/A | 100 | 90 |
| Al3+ | µg/l | 2 | N/A | 160 | 50 |
When the dolphins arrived in the Delfinaario on March 31, 1985, the bio-filter had not yet been conditioned properly. This was due to a delay in the building because of a long period of severe frost. In addition to this, due to the rapid blocking of the bio-plates, combined with the absence of an overflow possibility for the bio-filter tank, the flow over the bio-filter had to be reduced during the first weeks to less than 180 m3/hr. This resulted in a further delay of the conditioning of the filter. Once the overflow was installed, the flow could be increased and the filter rapidly started performing. In the meantime the level of ammonia (NH4-N) had risen to more than 11 mg/l. Shortly after the increase of the flow this level dropped dramatically, followed about 2 weeks later by a drop in the nitrite (NO2-N) levels. These levels have been very low since.
The average levels for 1986 for both are :
Both values are well below the maximum values for aquarium management, given by Spotte (1979a): 100 µ/l for both. He adds that these are rather conservative maxima, since even in fish toxic reactions do not occur until much higher concentrations.
As expected, the value of nitrate (NO3-N) increases continuously. The average increase over 1986 is 0.617 mg/l/day, representing roughly 70% of the nitrogen added in the form of food. The concentration at the end of 1986 was 400 mg/l. This is fairly high.
Although there are no toxic reactions to nitrate recorded even at considerably higher concentrations, from a management point of view it would be good to attempt to remove it. The obvious way to do this would be the installation of an anaerobic filter for denitrification.
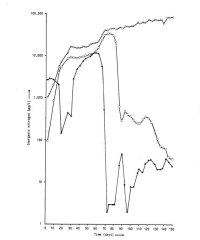
Nitrogen levels during the first weeks of operation of the biological filter.
Throughout 1986, the level of NH4-N was 37 ± 19 µg/l
and the level of NO2-N was 81 ± 22 µg/l.
NO3-N increased steadily at a rate of 0.616 mg/l/day.
Day 0 is March 31st, 1985, the day of the arrival of the dolphins at the Delfinaario.
The removal of carbon from the water seems to be very efficient. The concentration DOC (Dissolved Organic Carbon) has been shown to make up more than 90% of the total organic carbon (TOC) in the system by an independent laboratory. DOC levels in the Delfinaario have hardly changed as the following values show :
In chlorinated marine mammal pools the TOC increases constantly (Spotte and Adams, 1979). In this system all material that is biodegradable is broken down by micro-organisms and the rest can be removed through the foam fractionators.
The level of phosphate in the water had been rising constantly, but recently this increase slowly stopped. This is what is normally found in aquarium situations (Spotte, 1979b). Over the first half year of 1986 an increase was found of 0.049 mg/l/day (r=0.95) while over the second half of the year the increase is less obvious and also less linear : 0.010 mg/l/day (r=0.75). The concentration at the end of the year was a little over 20 mg/l. This is somewhat higher than would be expected in an aquarium (Spotte, 1979b). The reason for this is unknown. Possibly some complexing occurs, that prevents precipitation of phosphate with calcium and magnesium.
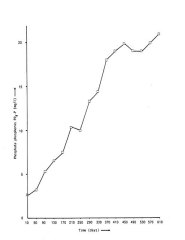
Concentration of phosphate phosphorus PO4-P. The concentration has remained constant at around 20 mg/l since the end of 1986. Day 0 is March 31st, 1985, the day of the arrival of the dolphins at the Delfinaario
In this system, no disinfecting agent, such as chlorine, has been added to the water. In part I of this paper (Van der Toorn, 1987) it was already pointed out that this might not be necessary (see also Wickins and Helm, 1981), since the ciliates present in the bio-filter graze on bacteria, and diatoms, which are present in significant amounts, secrete substances that inhibit the growth of bacteria (Aubert and Pesando, 1969).
Samples of the pool water have been analysed for bacterial contents usually twice weekly. The results of these tests for 1986 are shown in the graph below. From this it is clear that most samples had coliform counts, that were well below 100 colonies per ml. There were occasional high coliform counts. These were randomly distributed over the year and showed no correlation with for instance weather conditions, water temperature or any other factors. Apart from the coliform counts, the samples were analyzed for total bacterial contents, in number of colonies per ml. The results of these tests were highly variable. There was no correlation between the total bacterial count and the coliform count.
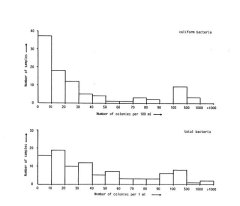
Results of the bacterial tests in 1986. The coliform counts are in number of colonies per 100 ml, the total bacteria counts in number of colonies per 1 ml. The total number of samples analyses N=95.
One of the most obvious features of the water in the Delfinaario is its deep green colour. This colour already existed when the dolphins arrived in Tampere and can therefore most likely not be attributed to organic matter. This has been confirmed by the measurements of several parameters. One of the parameters that has been measured for quite some time was the organic colour of the water, expressed in mg Pt/l. This value has constantly been and still is around 5 mg Pt/l. The increase of organic matter due to the arrival of the dolphins did not effect this value at all. The TOC values, which have not increased at all, confirm this. Also recently the concentration of chlorophyl has been measured. That concentration was 0.2 µg/l, which is very low if one compares it to the concentrations in the lakes around Tampere which range from 2 to over 30 µg/l (S.Näsi,pers.comm.). Postma (1983) recorded 25 µg/l or more for the Ems-Dollard estuary.
The explanation should therefore be sought in inorganic sources. Probably it is a combination of high concentrations of copper and manganese. Both are present in a rather high concentration in the town water supply of Tampere, which has a greenish colour. Further addition of these metals as contamination in the salts has increased the levels even more. Combination of these metals with organic molecules, or other ways of complexing, may prevent them from precipitation, thus keeping them in the solution.
1) The Photozone equipment was supplied by Ionisation Europe B.V., P.O.Box 53, 4240 CA Arkel, the Netherlands.
 Previous page
Previous page |
 Next page
Next page |
 Back to the Online Papers Page
Back to the Online Papers Page |
 Back to the Publications Page
Back to the Publications Page |